|
8/30/2023 0 Comments Viscosity of air centipoise The viscosity of water decreases smoothly from the freezing point while the density remains essentially constant except for the small maximum in density near 4☌. Online calculator, figures and tables with dynamic (absolute) and kinematic viscosity for air at temperatures ranging -100 to 1600☌ (-150 to 2900☏) and at pressures ranging 1 to 10 000 bara (14.5. From the table above - to convert from centipoise (cP). In other words, as it gets hotter, gases like air feel a bit stagnant than. so that at higher temperatures, the viscosity of gases goes high, too. We can express dynamic viscosity in millipascalssecond (mPas) or centipoise (cP) where 1 mPas 1 cP. In cgs units the unit is called the centistoke. Example - Converting Dynamic Viscosity from centipoise to lb/ft s. This water viscosity will help you determine the viscosity of water at. The kinematic viscosity is the dynamic viscosity divided by the density of the fluid. Water is a liquid at standard ambient temperature and pressure, but it often co-exists on Earth with its solid state, ice, and gaseous state, steam (water vapor). The unit centiPoise = 0.001 Pa s and is is sometimes written mPa s. A water molecule contains one oxygen and two hydrogen atoms that are connected by covalent bonds. These viscosities are at 20☌ except for the blood and blood plasma which are at body temperature, 37☌, and for steam which is at 100☌. The Poise is used in the table because of its more common usage. More commonly used is the dyne sec/cm 2 which is called Poise. 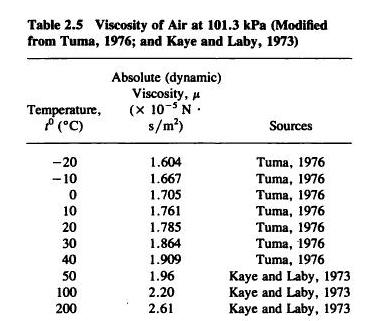
Viscosity has the SI units Pascal seconds (Pa s) which is called the Poiseuille. Skip to main content An official website of the United States government. Viscosity of Liquids and Gases Viscosity of Liquids and Gases Liquids Dynamic Viscosity (centiPoise) Kinematic Viscosity (centistoke) Density gm/cm 3: 2: 1.6735: 1.6736: 0.9999: 3: 1.619: 1.6191: 1: 4: 1.5673: 1.5674: 1: 5: 1.5182: 1. Tables of recommended values for the viscosity of common gases as a function of temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
